Galaxy DNA-Seq Tutorial
https://docs.rc.uab.edu/
Please use the new documentation url https://docs.rc.uab.edu/ for all Research Computing documentation needs.
As a result of this move, we have deprecated use of this wiki for documentation. We are providing read-only access to the content to facilitate migration of bookmarks and to serve as an historical record. All content updates should be made at the new documentation site. The original wiki will not receive further updates.
Thank you,
The Research Computing Team
Galaxy DNA-Seq Tutorial
Linking to data
Link in the Mark Pritchard Vaccinia virus data set.
- Start with a blank history, there should be no numbered items on the right hand side of the pane. Otherwise create a new history.
- Select "Shared Data" from the top of the screen to bring up the Shared Data screen
- Select Data Library
- Select "Pritchard Vaccinia WR FastQ Files" from the alphabetically sorted list
- Click on the subfolder top box to select all 6 files
- Select "import to current history"
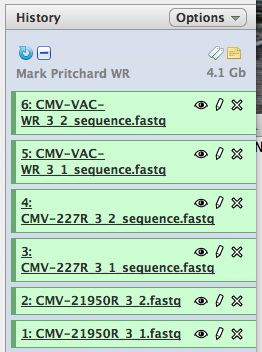
- Click on "Analyze Data" from the upper main menu. It should bring up the main page and your history pane should now look like the image below.
- Notice that with just 3 viruses are already over 4 GB of data.
Formatting and Grooming Data
- Your data is often NOT what you expect
FastQ Format
- FastQ format can mean many things, see the wikipedia entry on FastQ format
- Ilumina FastQ format also not informative, it changes
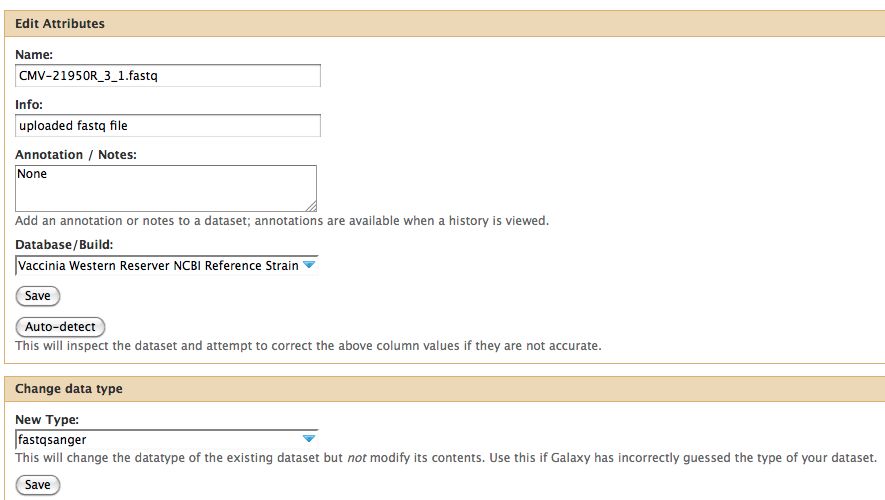
- Click on the pencil to icon in one of the virus images to pull up the attributes, your screen should look a bit like this:
- In Galaxy the expected data type of the galaxy tool must match EXACTLY with the data type in your history pane, otherwise the option to use that particular piece of data will not appear in the tool's drop down menu for data selection.
- Galaxy requires that everything go into Sanger format to be used. If you know your data is in sanger format, select fastqsanger for your data type. If it is not in that format, select fastq and run the FastQ Groomer.
- FastQ groomer is very useful, but slow
Reference Genome
- Ensure the correct reference genome is selected
- WARNING - seeing the reference genome doesn't mean it is there for using
- The current list of genomes available at UAB is here
- Used for a variety of tools, it can be overridden earlier
Assessing the quality of the data
We will use a number of different tools from the "NGS: QC and manipulation" drop down menu. Try processing the FastQ files with:
- FastQC
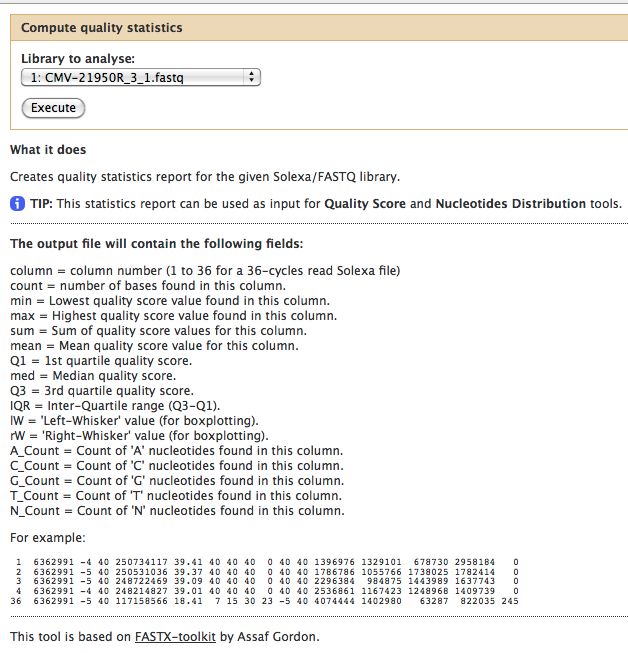
- Compute Quality Statistics
FastQC gives an attractive visual output and will flag potential problems.
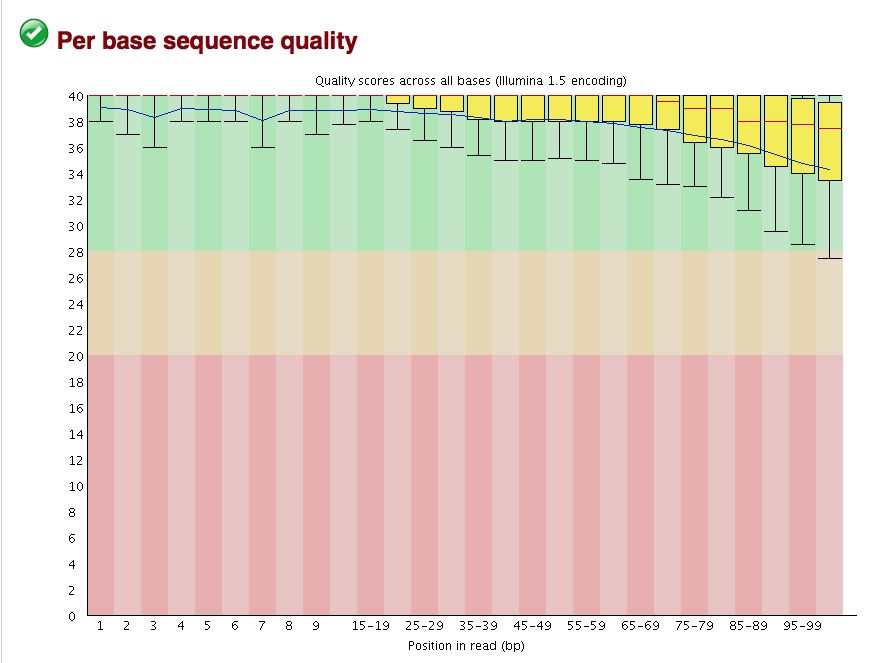
Take a look at the other areas that show up as quality issues. Can any of these be a problem for us?
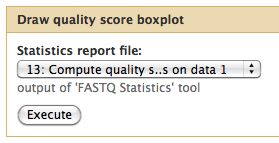
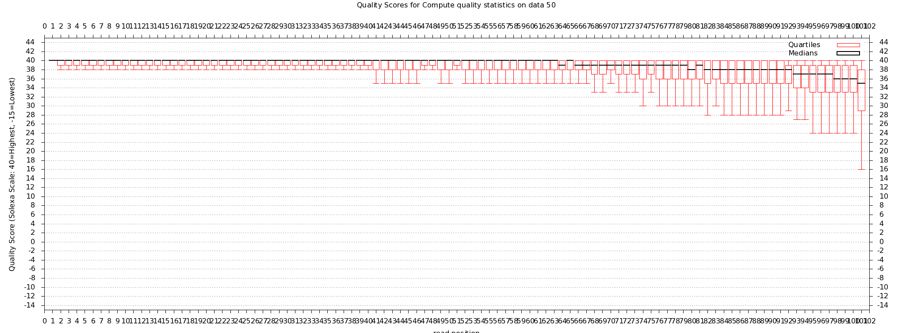
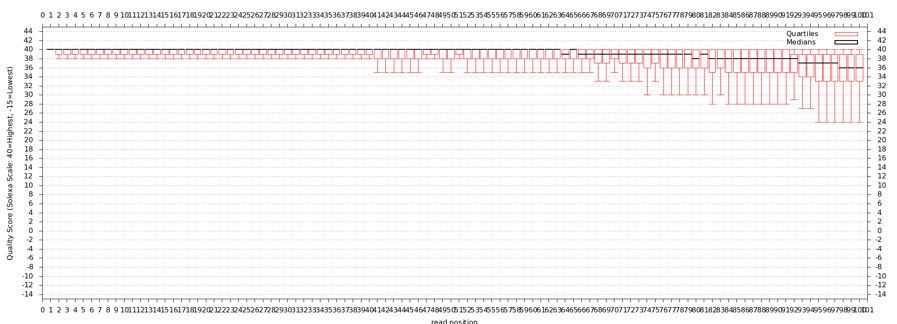
The quality statistics computing by the 2nd operation can be used for another box and whiskers plot to look at the read quality on a base by base basis. Select:
- "NGS: QC and manipulation" -> Draw Quality Score Boxplot
Performing cleanup
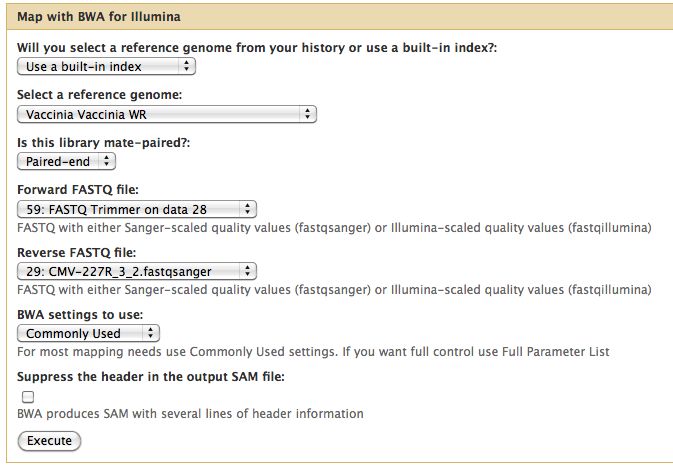
Short read alignment to reference genome using BWA
BAM Conversion
The output of many mapping programs is in SAM format and must be converted to the binary format. The conversion is lossless.
Variant Analysis
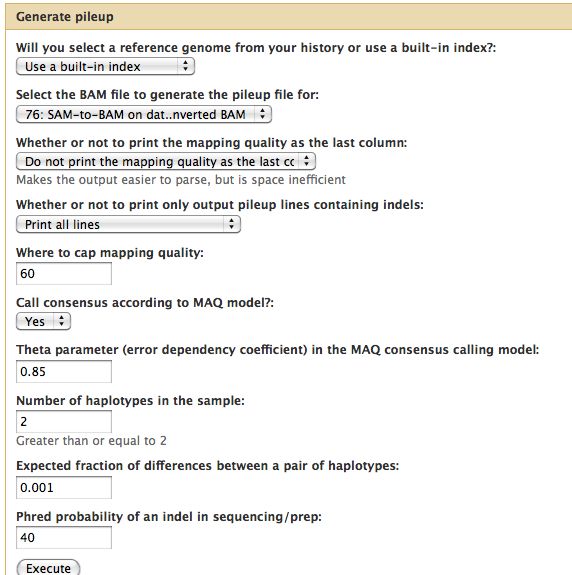
Samtools
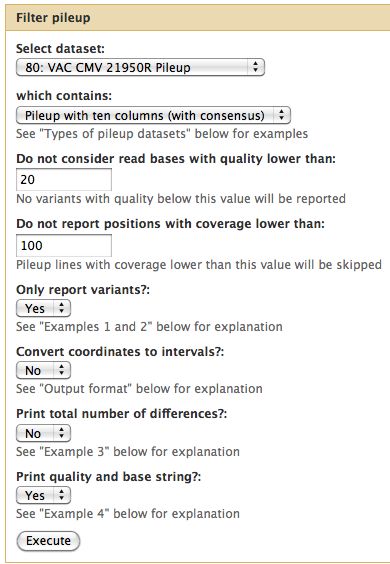
- Essential toolset, performs a variety of functions including generation of a "pileup" file.
Other Variant Detection Approaches
- GATK - Emerging as the new best practise to call variants, not integrated into Galaxy just yet.
- SNPEff
- PATRIC, annotation sites
SNPEff and Variant Summation
Summarizing variants and effects of SNPs.
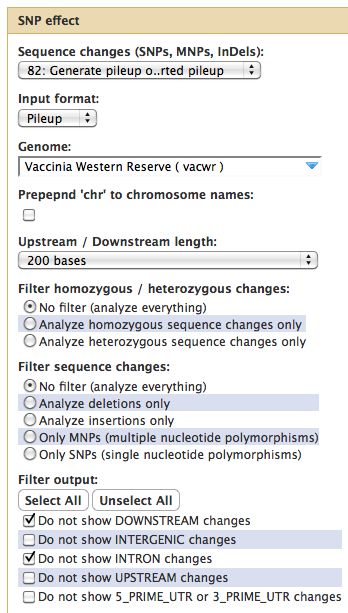
- Reference genome must be in SNPEff, need GFF3 file of your genome annotation
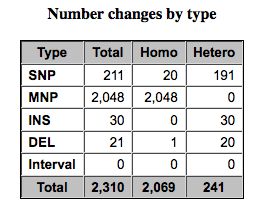
- SNPEff looks at heterozygous and homozygous SNPs, MNPs
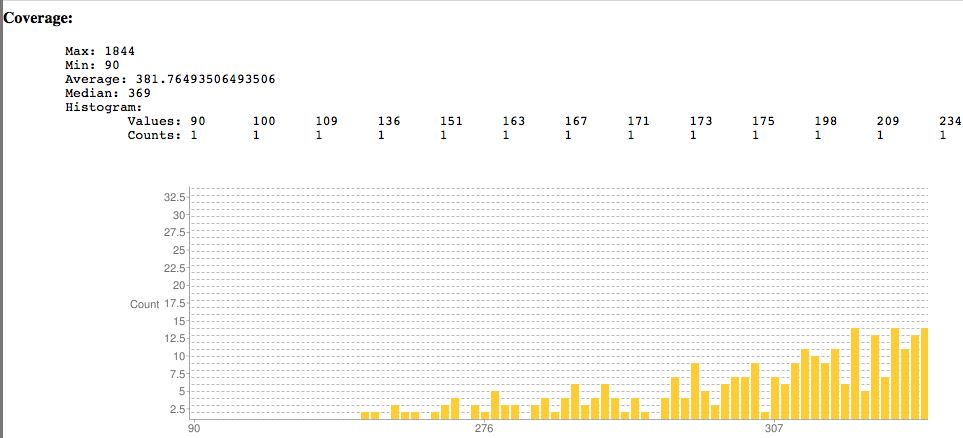
- Plots coverage, indel sizes
Running SNPEff
Results from SNPEff
- Only as good as your genome annotation file
- Introns in viruses?
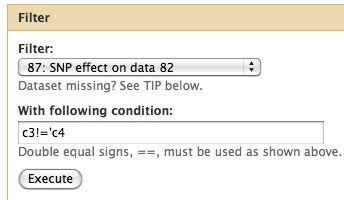
Locating Relevant SNPs versus Control